Trending...
- New Home of the Month: Spacious Luxury Meets Modern Design in The Bristol at Heritage at Manalapan - 126
- Joseph Nybyk aka Neibich of Gilbert, Arizona
- Don Barnhart Extends Hit Las Vegas Residency At Delirious Comedy Club Inside Silver Sevens
NRx Pharmaceuticals (N A S D A Q: NRXP) $NRXP: Dual FDA Pathways, and an Expanding AI-Driven Treatment Ecosystem Position NRXP at the Forefront of a Multi-Billion-Dollar Mental Health Transformation
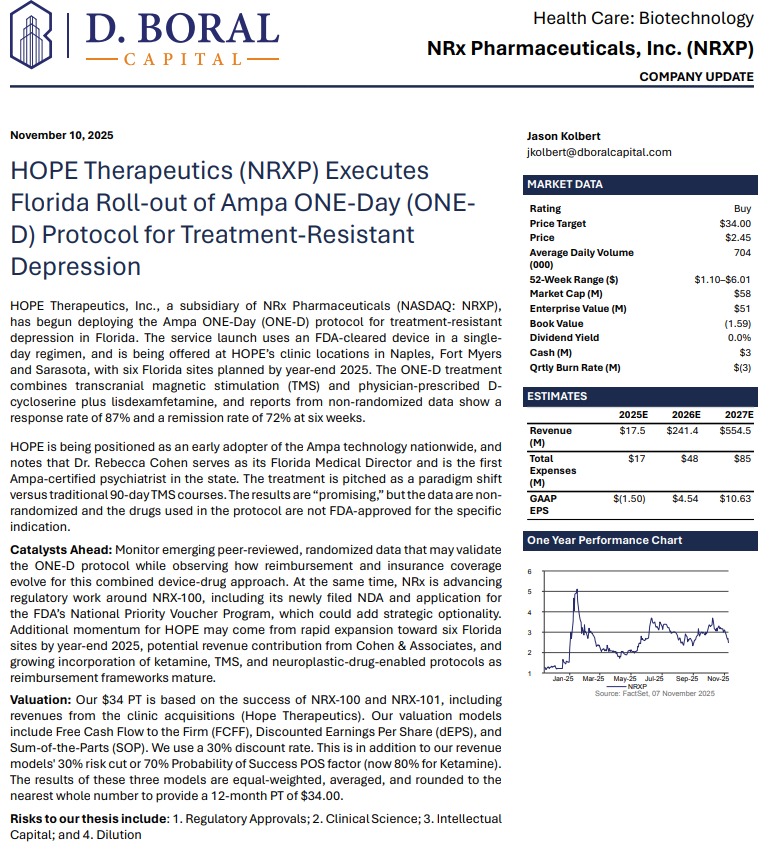
MIAMI - nvtip -- NRx Pharmaceuticals (N A S D A Q: NRXP) is rapidly emerging as a high-impact, late-stage biotech story transitioning into a nationally aligned healthcare platform, as a powerful convergence of regulatory clarity, federal policy support, and technological innovation reshapes both its timeline and total addressable market.
The company's latest developments place it squarely at the intersection of urgent public health need and government-backed acceleration, following the April 18, 2026 Presidential Executive Order aimed at fast-tracking treatments for serious mental illness, including psychedelic and breakthrough therapies.
This directive is expected to expand the use of real-world evidence, accelerate FDA review pathways, and introduce priority mechanisms such as Commissioner's National Priority Vouchers—one of which NRx has already applied for. The result is a meaningful shift in the regulatory landscape, positioning companies like NRx to benefit from faster approvals, increased prioritization, and national-level visibility.
FDA Alignment Transforms the Timeline
At the core of the investment thesis is a rare and highly material regulatory breakthrough. Following direct engagement with the FDA, NRx confirmed that its lead candidate, NRX-100 (preservative-free ketamine), can proceed toward a New Drug Application using existing clinical trial data combined with real-world evidence—eliminating the need for additional trials.
This positions the company to target an NDA submission as early as June 2026 while simultaneously expanding the proposed indication to include a broader population of patients suffering from treatment-resistant depression with suicidality.
To support this filing, NRx is preparing to submit data from more than 70,000 patients, creating one of the most robust real-world datasets in the psychiatric treatment space.
Key regulatory positioning now includes:
Dual FDA Catalysts Create a Rare Parallel Opportunity
More on nvtip.com
While the NDA pathway represents transformational upside, NRx is simultaneously advancing a second regulatory track that may generate nearer-term revenue.
The company's Abbreviated New Drug Application for preservative-free ketamine in anesthesia has already achieved bioequivalence confirmation and near-final labeling alignment, with anticipated FDA action in the second half of 2026.
This dual-track structure creates a compelling dynamic:
Few companies at this stage possess two aligned regulatory catalysts with overlapping timelines.
From Biotech to Platform: Expanding Into Neurotechnology and AI
NRx is no longer operating as a single-asset biotech. It is building an integrated platform that combines pharmaceuticals, neurostimulation, and artificial intelligence.
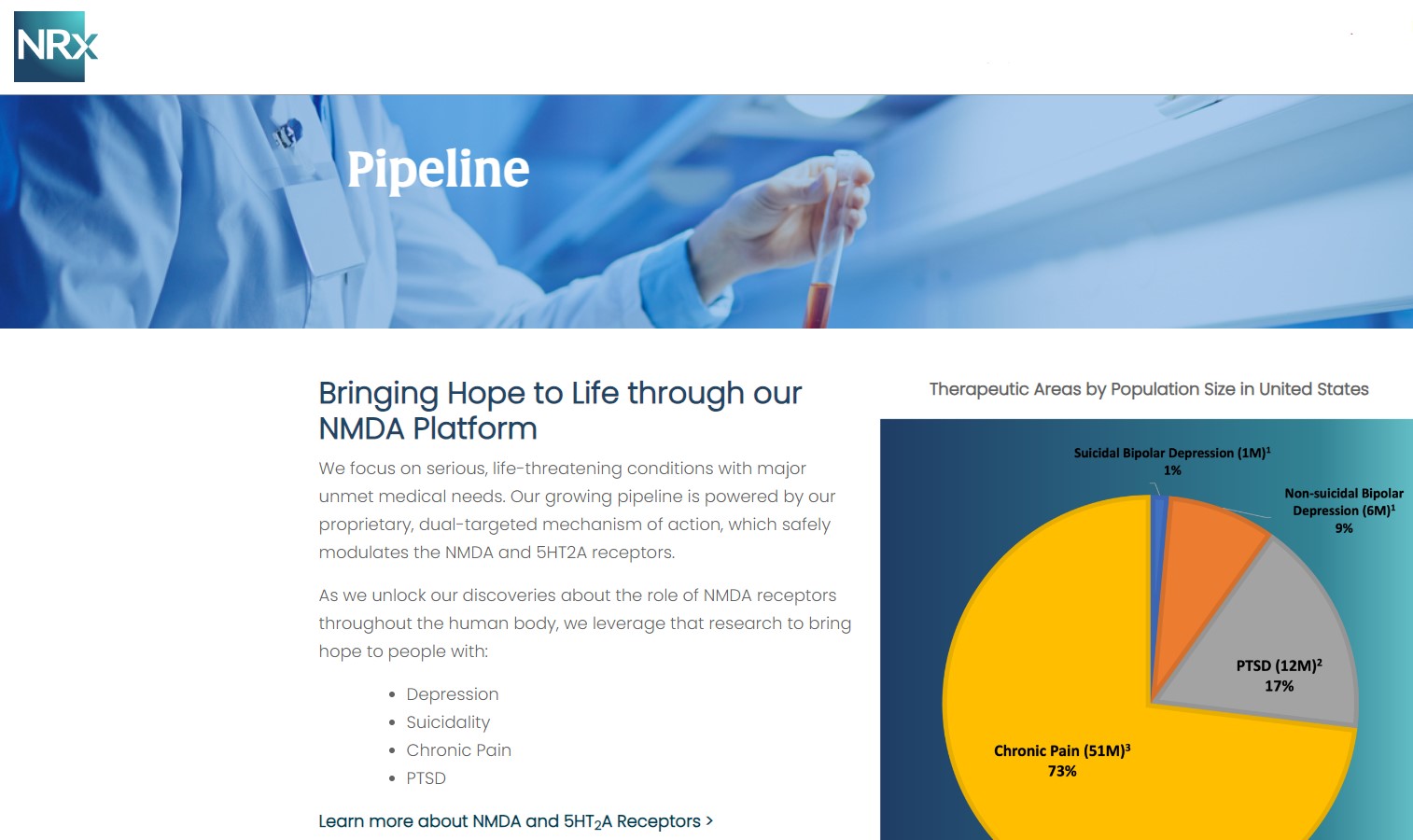
Its Breakthrough Therapy-designated candidate, NRX-101, is being developed as a maintenance therapy for suicidal bipolar depression and chronic pain, designed to work synergistically with neuroplastic treatment approaches.
That strategy is now expanding into advanced neurotechnology through the formation of NRx Defense Systems, a subsidiary focused on robotic-enabled Transcranial Magnetic Stimulation (TMS) combined with neuroplastic drug therapy.
This initiative targets military personnel and first responders—populations with significantly elevated rates of PTSD and depression—and introduces the potential for government-backed, non-dilutive funding channels.
Technology and platform expansion highlights:
Commercial Infrastructure Already in Motion
Importantly, NRx is not waiting for regulatory approvals to build its business. Through its HOPE Therapeutics subsidiary, the company has established a growing network of interventional psychiatry clinics delivering neuroplastic therapies, including ketamine and TMS.
These clinics are already generating revenue and are supported by a combination of government programs, insurance reimbursement, and private-pay patients.
In parallel, the company has partnered with neurocare AG to expand a nationwide treatment network that includes:
This infrastructure provides a ready-made commercialization channel upon approval.
More on nvtip.com
AI Integration Adds a New Layer of Precision Medicine
NRx is further differentiating itself through the integration of artificial intelligence into patient care.
Through its partnership with Emobot, the company is deploying a passive monitoring platform—often described as a "Depression Thermometer"—that continuously evaluates patient condition via smartphone-based data collection.
This enables earlier intervention, improved treatment optimization, and a scalable model for managing chronic mental health conditions.
Financial Position Strengthens Execution Capability
The company has also made meaningful progress in strengthening its financial foundation.
Recent disclosures highlight reduced operating expenses, the elimination of balance sheet debt, and sufficient capital to support operations through 2026.
This positions NRx to execute on its regulatory and commercial strategy without immediate financing pressure.
A Market Defined by Urgency and Scale
NRx is targeting one of the most significant unmet needs in healthcare. More than 13 million Americans report seriously considering suicide each year, while treatment-resistant depression and PTSD continue to rise globally.
With ketamine-based therapies demonstrating rapid clinical impact and federal policy now accelerating access to innovative treatments, the demand environment is both immediate and expanding.
Final Take: A Convergence Story with Multiple Inflection Points
NRx Pharmaceuticals is no longer simply advancing a drug candidate—it is building a multi-dimensional platform at the intersection of biotech, AI, neurotechnology, and national healthcare policy.
With:
NRx now represents a rare type of opportunity in the market:
A company where regulatory clarity, government support, and commercial execution are all advancing at the same time
For aggressive, forward-looking investors, NRXP is not just approaching a catalyst—
$NRXP is entering a phase where multiple catalysts are beginning to compound.
For more information on $NRXP visit: https://www.nrxpharma.com/ and https://compasslivemedia.com/case-study/nrx-pharmaceuticals/
Company Contact
NRx Pharmaceuticals, Inc. (N A S D A Q: NRXP)
Matthew Duffy – Chief Business Officer
Phone: (484) 254-6134
Email: mduffy@nrxpharma.com
DISCLAIMER: https://corporateads.com/disclaimer/
Disclosure listed on the CorporateAds website
The company's latest developments place it squarely at the intersection of urgent public health need and government-backed acceleration, following the April 18, 2026 Presidential Executive Order aimed at fast-tracking treatments for serious mental illness, including psychedelic and breakthrough therapies.
This directive is expected to expand the use of real-world evidence, accelerate FDA review pathways, and introduce priority mechanisms such as Commissioner's National Priority Vouchers—one of which NRx has already applied for. The result is a meaningful shift in the regulatory landscape, positioning companies like NRx to benefit from faster approvals, increased prioritization, and national-level visibility.
FDA Alignment Transforms the Timeline
At the core of the investment thesis is a rare and highly material regulatory breakthrough. Following direct engagement with the FDA, NRx confirmed that its lead candidate, NRX-100 (preservative-free ketamine), can proceed toward a New Drug Application using existing clinical trial data combined with real-world evidence—eliminating the need for additional trials.
This positions the company to target an NDA submission as early as June 2026 while simultaneously expanding the proposed indication to include a broader population of patients suffering from treatment-resistant depression with suicidality.
To support this filing, NRx is preparing to submit data from more than 70,000 patients, creating one of the most robust real-world datasets in the psychiatric treatment space.
Key regulatory positioning now includes:
- NDA pathway for NRX-100 with no additional trials required
- Fast Track designation already in place
- Real-world evidence accepted as part of the approval framework
- Expanded label strategy significantly increasing market potential
Dual FDA Catalysts Create a Rare Parallel Opportunity
More on nvtip.com
- J&J Exterminating Celebrates 65th Anniversary and Unveils Strategic Vision at Annual Team Meeting
- Tru by Hilton El Paso Airport Opens to Guests
- Zenylitics Announces Leadership Transition to Continue Accelerated Growth
- Wellness Technology Distributor Helping People Set Up Wellness Center Businesses
- Christian Apocalyptic Thriller Explores Biblical Prophecy, Global Technology, & the Rise of the Ant
While the NDA pathway represents transformational upside, NRx is simultaneously advancing a second regulatory track that may generate nearer-term revenue.
The company's Abbreviated New Drug Application for preservative-free ketamine in anesthesia has already achieved bioequivalence confirmation and near-final labeling alignment, with anticipated FDA action in the second half of 2026.
This dual-track structure creates a compelling dynamic:
- Near-term commercialization potential via ANDA approval
- Longer-term, high-value expansion via NDA approval
Few companies at this stage possess two aligned regulatory catalysts with overlapping timelines.
From Biotech to Platform: Expanding Into Neurotechnology and AI
NRx is no longer operating as a single-asset biotech. It is building an integrated platform that combines pharmaceuticals, neurostimulation, and artificial intelligence.
Its Breakthrough Therapy-designated candidate, NRX-101, is being developed as a maintenance therapy for suicidal bipolar depression and chronic pain, designed to work synergistically with neuroplastic treatment approaches.
That strategy is now expanding into advanced neurotechnology through the formation of NRx Defense Systems, a subsidiary focused on robotic-enabled Transcranial Magnetic Stimulation (TMS) combined with neuroplastic drug therapy.
This initiative targets military personnel and first responders—populations with significantly elevated rates of PTSD and depression—and introduces the potential for government-backed, non-dilutive funding channels.
Technology and platform expansion highlights:
- Development of robotic, AI-guided TMS systems
- Integration with NRX-101 for enhanced neuroplastic response
- Prototype expected mid-2026
- Clinical data showing up to 87% response rates in related therapies
Commercial Infrastructure Already in Motion
Importantly, NRx is not waiting for regulatory approvals to build its business. Through its HOPE Therapeutics subsidiary, the company has established a growing network of interventional psychiatry clinics delivering neuroplastic therapies, including ketamine and TMS.
These clinics are already generating revenue and are supported by a combination of government programs, insurance reimbursement, and private-pay patients.
In parallel, the company has partnered with neurocare AG to expand a nationwide treatment network that includes:
- 20+ clinical sites
- More than 400 installed TMS systems
This infrastructure provides a ready-made commercialization channel upon approval.
More on nvtip.com
- Avery Burton Foundation Announces 2026 Student Scholarship Award Winners
- The Hidden Price Of Lost Property In UK Schools
- Milton Collier, CEO & President of Freight Broker 911, Eliminates the #1 Barrier to Entry in Logistics: Announces 100% Free Freight Broker Training
- FDA-Cleared AI Neuropsychiatry Platform, Million-Dose Ketamine Manufacturing and Presidential Psychedelic Initiative Drive Growing Momentum for NRXP
- AI Velocity Trading Launches Institutional-Grade Algorithmic Engine for Retail Investors
AI Integration Adds a New Layer of Precision Medicine
NRx is further differentiating itself through the integration of artificial intelligence into patient care.
Through its partnership with Emobot, the company is deploying a passive monitoring platform—often described as a "Depression Thermometer"—that continuously evaluates patient condition via smartphone-based data collection.
This enables earlier intervention, improved treatment optimization, and a scalable model for managing chronic mental health conditions.
Financial Position Strengthens Execution Capability
The company has also made meaningful progress in strengthening its financial foundation.
Recent disclosures highlight reduced operating expenses, the elimination of balance sheet debt, and sufficient capital to support operations through 2026.
This positions NRx to execute on its regulatory and commercial strategy without immediate financing pressure.
A Market Defined by Urgency and Scale
NRx is targeting one of the most significant unmet needs in healthcare. More than 13 million Americans report seriously considering suicide each year, while treatment-resistant depression and PTSD continue to rise globally.
With ketamine-based therapies demonstrating rapid clinical impact and federal policy now accelerating access to innovative treatments, the demand environment is both immediate and expanding.
Final Take: A Convergence Story with Multiple Inflection Points
NRx Pharmaceuticals is no longer simply advancing a drug candidate—it is building a multi-dimensional platform at the intersection of biotech, AI, neurotechnology, and national healthcare policy.
With:
- Federal acceleration of mental health treatment approvals
- FDA-aligned NDA pathway requiring no new trials
- Parallel ANDA approval opportunity
- Breakthrough Therapy designation for NRX-101
- Expanding clinic network generating real-world revenue
- AI-driven patient monitoring and treatment optimization
- Entry into defense-focused neurotechnology
NRx now represents a rare type of opportunity in the market:
A company where regulatory clarity, government support, and commercial execution are all advancing at the same time
For aggressive, forward-looking investors, NRXP is not just approaching a catalyst—
$NRXP is entering a phase where multiple catalysts are beginning to compound.
For more information on $NRXP visit: https://www.nrxpharma.com/ and https://compasslivemedia.com/case-study/nrx-pharmaceuticals/
Company Contact
NRx Pharmaceuticals, Inc. (N A S D A Q: NRXP)
Matthew Duffy – Chief Business Officer
Phone: (484) 254-6134
Email: mduffy@nrxpharma.com
DISCLAIMER: https://corporateads.com/disclaimer/
Disclosure listed on the CorporateAds website
Source: CorporateAds
0 Comments
Latest on nvtip.com
- Color Card Administrator Highlights Growing Enterprise Demand for Workflow Orchestration in Enterprise Business Card Governance
- Tennessee Laws Lead with Psychotropic Drug Testing in Mass Shooting Cases and Comprehensive Reporting: CCHR Urges Nationwide Adoption
- Curious About Mensa? DFW Event Offers a 1-Day Immersion
- Buzzblender Announces Launch of Simple Hotel Mode for Android and Upcoming Video Wall Support for Samsung Professional Displays
- How Strategic WooCommerce Development and Digital Marketing Helped a Fashion Ecommerce Business Increase Revenue by 3X
- VIV Welcomes Residents to St. Petersburg's EDGE District
- Evocative Joins the Independent Data Centre Network (IDCN) as Primary USA Operator
- Medical Experts Highlight the Importance of Second Opinions in Death Investigations
- Joseph Nybyk aka Neibich of Gilbert, Arizona
- Don Barnhart Extends Hit Las Vegas Residency At Delirious Comedy Club Inside Silver Sevens
- Omnitronics Unveils 100% Software omniGateDMR and omniGateP25 RoIP Gateways
- KRE PRIME Launches Adaptive Convertible Jumpsuit
- Jeff Miller for Clark County Clerk: Campaign Signals Strong Early Voting Momentum in Las Vegas
- USA Med Bed Helping Home Care Patients with Refurbished Hill Rom Hospital Beds
- Sobreseimiento de Nicolás dos Santos y Jorge Méndez expone demandas millonarias a Paraguay y boicot a la Hidrovía
- CAPHRA warns Southeast Asia not to repeat Australia's nicotine policy failure
- Milo3D.ai Launches Free AI 3D Model Generator That Turns Text and Images Into Game-Ready 3D Assets in Seconds
- UK Financial Ltd Executes 100% Success Rate on All ERC-3643 Transfers to Coin Holders of MayaCat Regulated Security Token and Maya Preferred PRA
- Blank Space: The Unofficial Taylor Swift Tribute Brings Eras Tour Magic To Cities Across America
- Love Must Be the Guide: Live Good Shares a Message of Humanity, Compassion and Hope